1, followed another judge's decision in 2017 to dismiss the suit without prejudice, finding that their state-law claims had been pre-empted by federal law.Īccording to court documents, medical products like St. The 33-page memorandum opinion, issued Feb. Jude failed to warn her and her doctors of the risks associated with its product. However, Burke said that Freed's revised complaint had laid out an initial case that the device was dangerous, and granted the Freed one more chance to show that St. Though the procedure was intended to be permanent, Freed said she had the device removed less than one year later, after complaining that it had caused painful electric shocks and burning. Jude had failed to live up public guarantees regarding the safety of its spinal cord simulator, which was surgically implanted in her during a procedure at Newark's Christiana Hospital in 2014. must defend part of a products liability suit claiming the Minnesota-based medical device maker had negligently manufactured a spinal cord stimulator that was implanted in a Maryland woman. Many patients suffering from chronic pain may ultimately need an MRI in the future.A Delaware federal magistrate judge ruled last week that St. Past research has suggested that the condition can cost the American population an estimated 515 million workdays annually and generates upwards of 40 million visits to physicians each year. Our new labeling for the Proclaim Elite SCS system ensures that patients can receive BurstDR stimulation while having the peace of mind knowing their SCS system can be safely scanned with diagnostic imaging.”Īccording to the Institute of Medicine, chronic pain affects more than 100 million Americans, an incidence rate which outpaces heart disease, cancer and diabetes combined. “Historically, the need for future MRI scans could act as a barrier to patients who may benefit from SCS therapy. Burton, M.D., medical director of neuromodulation and vice president of medical affairs at St. 
“Today, physicians treating patients with chronic pain need new therapy options that consistently put the patient first and ensure minimal disruption to their daily life,” said Allen W. Jude Medical designed many elements of the Proclaim system based on physician and patient feedback, such as removing the burden of regularly recharging the SCS system. The system’s Bluetooth wireless technology allows patients to control and adjust their SCS therapy with an Apple iPod touch mobile digital device. The Proclaim Elite SCS System was the first upgradeable, recharge-free system available to people in the U.S. Luke's University Health Network in Bethlehem, Pa. Falowski, chief of functional neurosurgery at St. Jude Medical’s innovations without fear of putting themselves and their implant at risk,” said Dr. “The addition of full-body MR Conditional labeling for the Proclaim Elite SCS system will be another critical benefit for my patients and will help me ensure those who may one day need an MRI can benefit from St. Physicians nationwide have begun to offer their patients access to BurstDR stimulation. Jude Medical’s proprietary BurstDR stimulation works differently from other stimulation designs, utilizing intermittent “burst” pulses designed to mimic the body’s natural nerve impulse patterns. BurstDR stimulation is a physician-designed form of SCS clinically proven to provide patients superior pain relief (as compared to tonic stimulation) by reducing their physical pain and addressing their emotional response to pain as measured by visual analogue scale (VAS) scoring. Jude Medical designed the Proclaim Elite SCS system to offer patients future approved upgrades through software updates without the need to surgically replace their device. Current and future patients who get the Proclaim Elite SCS system will now have access to both full-body MRI scans within approved parameters and BurstDR stimulation. “I am very grateful that I can now safely expand access to the superior therapeutic outcomes offered by BurstDR stimulation to my patients who may need a full-body MRI in the future.”įull-body MRI compatibility is the second major upgrade to the Proclaim Elite SCS system following the recent approval of BurstDR stimulation. 
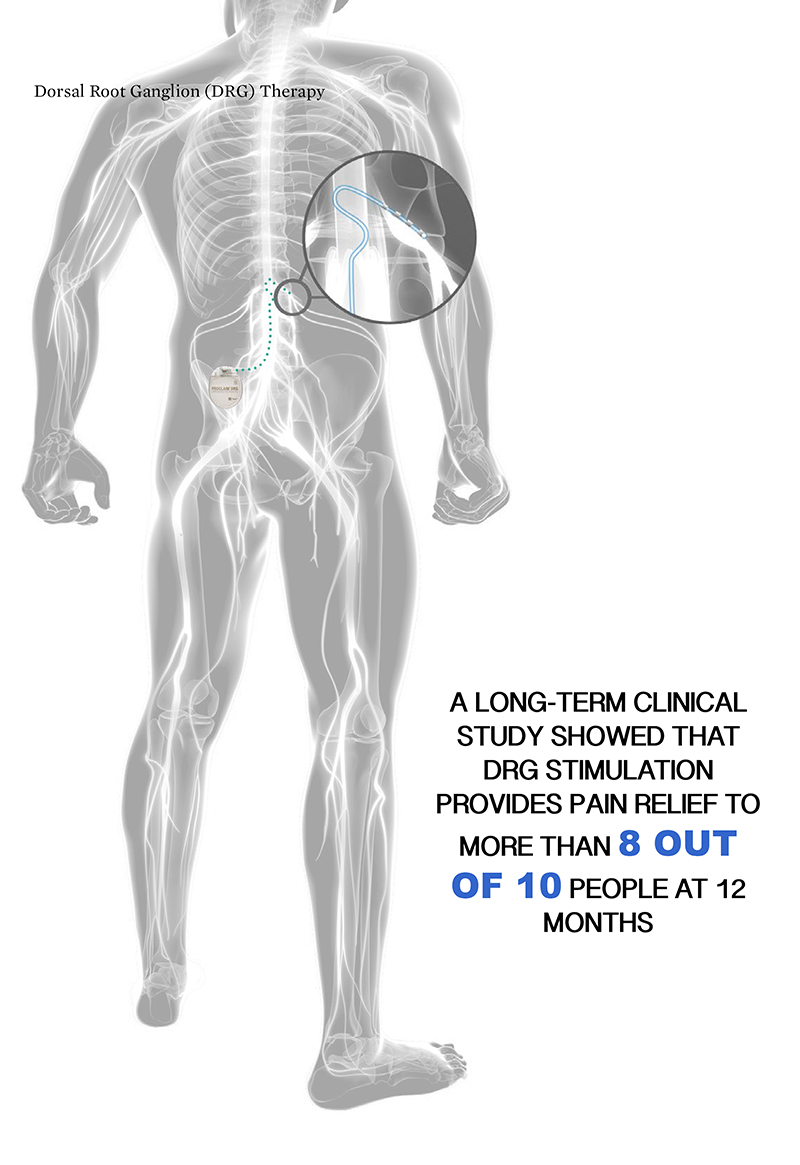
Yearwood of Comprehensive Pain & Rehabilitation in Pascagoula, Miss. “This MR Conditional labeling applies to both new patients and those already implanted with the upgradeable Proclaim Elite SCS System,” said Dr. With the latest labeling approval, patients who receive the Proclaim Elite SCS system can now undergo full-body magnetic resonance imaging (MRI) diagnostic scans within approved parameters and access the benefits of BurstDR stimulation therapy. Food and Drug Administration approval for full-body magnetic resonance (MR) Conditional labeling for the Proclaim Elite Spinal Cord Stimulation (SCS) System. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
